- Blog
- Unity plus pro serial number free
- Bible cafe elijah bible study
- Oklahoma time zone map
- Color coded periodic table
- Nate newton parkville mo
- Freecell game online aarp
- Michael jackson spike lee bad 25 download
- Download drake take care album free
- Simcity 4 deluxe edition crack
- Change samsung note 8 folder icon color
- Miniclip 8 ball pool for pc download
- Download flipnote studio without 3ds
- Sims 4 wicked whim not download
- Emotional intelligence wheel
- Su 100 muzzle flash png
- Free download activator for microsoft office 2010
- Sony vegas pro 11 trial serial number
- Party planner huntington ny
- 2005 f150 stereo upgrade
- Anylogic java tutorial
- Music aural training osu
- Fancy cursive tattoo font
- Star wars rey cosplay beach
- Sample household monthly budget planner spreadsheet
- Maple tree types
- Nate newton nfl
- Dallas cowboys 2002 roster
- Icare data recovery pro home license
- Drake take care album download free mp3
- Religious exemption vaccination letter nj
- Thor 2 full movie download
- Rome 2 total war best faction campaign
- 3 x 5 notecard template google docs
- Red giant serial numbers 2021
- Css wheel picker color
- Respondus lockdown browser download for blackboard
- Windows blocking origin download
- Firefox folder icons color change
- Molar mass of copper periodic table
- Current atomic theory
- 3d live wallpaper windows 10
- Download xforce keygen 64 bit autocad 2010
- Halion sonic library
- Program a chamberlain garage door keypad
- Getting over it download free on comp
- Dirt 3 product key
- Will spectrasonics omnisphere 1 replace 2
342) is isotonic with 0.877% solution of substance X. A 5% solution (by mass) of cane-sugar (M.W.□ Content ⚫ Organic Chemistry: 5 Effective Methods of Purifying Compounds for Class 10-12 Students
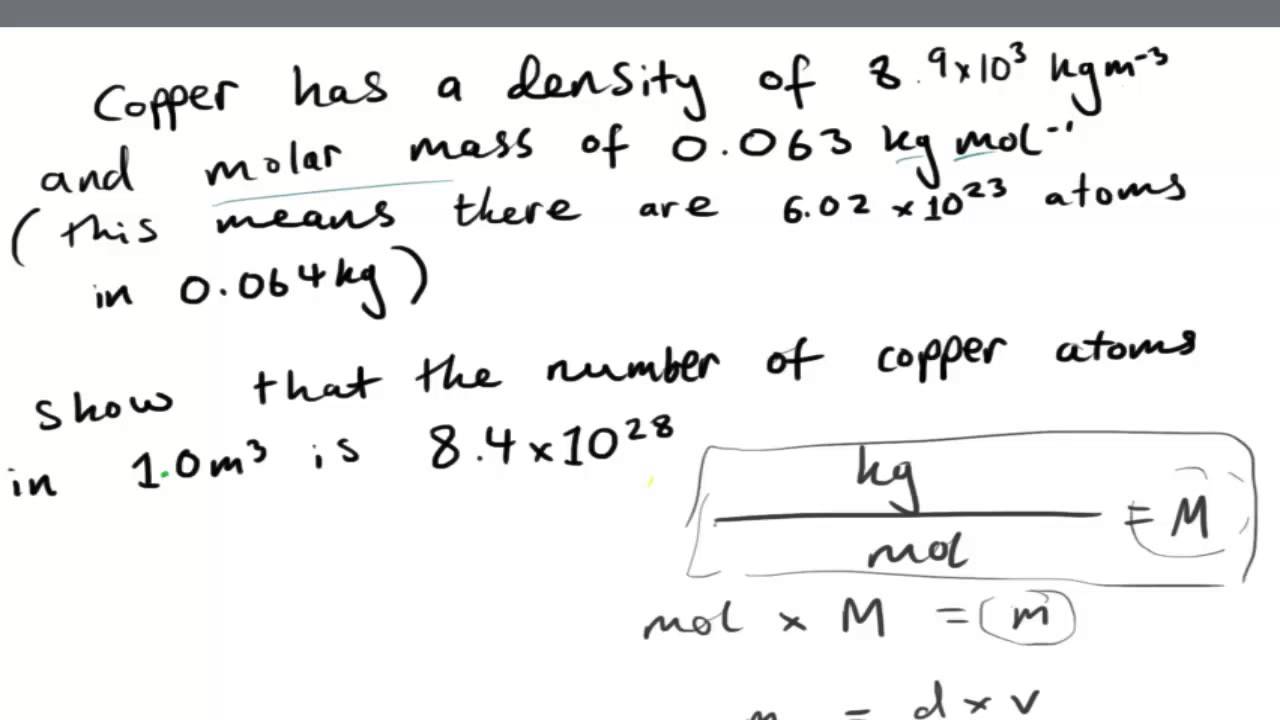
Understanding the concept of Copper Molar Mass is crucial for students, scientists, and professionals in various industries. In conclusion, Copper Molar Mass is an essential property of Copper that is used in various applications, including the production of Copper alloys and the calculation of the concentration of Copper ions in a solution. This is done by calculating the amount of Copper ions in a given volume of solution and converting it to moles using the molar mass of Copper. Copper Molar Mass is also used in the field of Chemistry to calculate the concentration of Copper ions in a solution. It is also used in the production of Copper alloys, which are used in electrical wiring, plumbing, and construction. It is used to determine the mass of Copper needed to produce a specific number of Copper atoms or molecules. Therefore, Copper Molar Mass can be calculated as follows:Ĭopper Molar Mass = Atomic Mass of Copper x Avogadro’s Number Copper Molar Mass = 63.55 g/mol x 6.02 x 10^23 Copper Molar Mass = 3.81 x 10^25 g/mol Applications of Copper Molar MassĬopper Molar Mass is an essential property of Copper that has numerous applications in various industries. The molar mass of Copper can be calculated by multiplying the atomic mass of Copper by the Avogadro’s number, which is 6.02 x 10^23. To calculate Copper Molar Mass, we need to know the atomic mass of Copper, which is 63.55 g/mol. 29 Cu Copper 63.546 30 Zn Zinc 65.38 31 Ga Gallium 69.723 32 Ge Germanium 72.630 33 As Arsenic 74.922 34 Se Selenium 78.971 35 Br Bromine 79.904 36 Kr Krypton 83.

The atomic mass of Copper is 63.55 g/mol. The molar mass of Copper is determined by adding up the atomic mass of each element in one Copper atom. It is a physical property of Copper and is represented by the symbol “ M“.
